NRTIs
Reviewed by: HU Medical Review Board | Last reviewed: May 2024 | Last updated: May 2024
Nucleoside reverse transcriptase inhibitors (NRTIs) are a class of drugs used to suppress HIV in the body. When NRTIs are used along with other HIV-fighting drugs, the treatment regimen is referred to as antiretroviral therapy (ART).1
NRTIs act on the reverse transcription portion of the HIV life cycle, where the virus transforms itself from RNA to DNA inside human host cells to carry out the rest of its replication process.1
The reverse transcription process
Viruses, like HIV, need human host cells to replicate. They cannot multiply on their own without human cells. When HIV particles, called virions, enter the body after a transmission event, the next main steps of the HIV lifecycle are as follows:2
- Binding
- Fusion
- Reverse transcription
- Integration
- Replication
- Assembly
- Budding
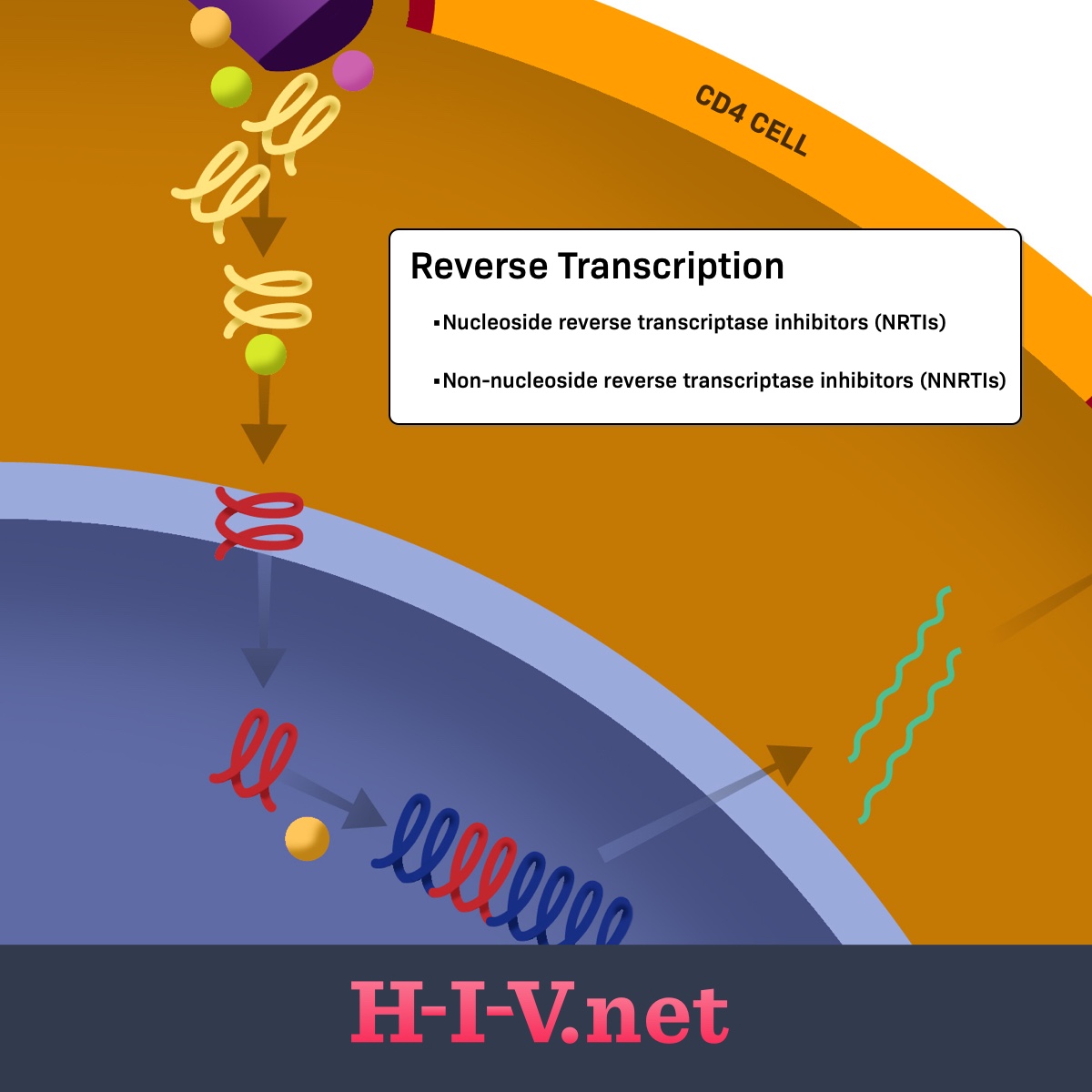
Figure 1. NRTIs target reverse transcription in the HIV life cycle
The first two steps of the life cycle focus on HIV getting into a human CD4 cell, or T cell, which is its target cell. Once inside, it needs to disassemble itself and begin the replication process. Normally in our cells, our body converts the information in our DNA to RNA (also called transcription) and then RNA to proteins. DNA and RNA are both forms of genetic material.2
HIV can go backward through this process to become DNA through a process called reverse transcription. HIV carries around a special enzyme called reverse transcriptase, which allows it to move backward from RNA to DNA and insert into the host cell’s DNA so it can continue to be replicated in its original form with the rest of our normal DNA. This special property of HIV is why it is classified as a retrovirus.2
Once HIV replicates itself, it can then reassemble into new virion particles that get released out of the cell and back into the bloodstream to infect new CD4 cells.2
How do NRTIs work?
The purpose of NRTIs is to stop the reverse transcription process. Normally, HIV uses its reverse transcriptase enzyme to build a strand of DNA from its original RNA form. To do this, it uses genetic building blocks called nucleosides that come from the host cell. These nucleosides are strung together one at a time, like beads on a string, to create the final DNA product.2
NRTIs act as nucleoside mimics that stop the DNA-building process. NRTIs look like normal nucleosides, except they’re missing a special chemical group on one side. Without this group, the reverse transcriptase enzyme cannot attach more nucleosides to the chain after them. This prevents the full string of DNA from being formed, preventing HIV RNA from making it through the reverse transcription process and into host DNA for further replication.2
Examples of NRTIs
NRTIs approved to treat HIV include:1,3-7
- Emtriva® (emtricitabine)
- Epivir (lamivudine)
- Retrovir (zidovudine)
- Viread® (tenofivir disoproxil fumarate)
- Ziagen (abacavir)
What are the possible side effects of NRTIs?
Side effects can vary depending on the specific drug you are taking. The most common side effects of NRTIs include:3-7
- Rash
- Headache
- Trouble sleeping (insomnia)
- Nausea, vomiting
- Tiredness
- Abdominal pain
- Dizziness
Many NRTIs have boxed warnings, the strictest warning from the US Food and Drug Administration (FDA). Before taking NRTIs, discuss with your doctor whether it is right for you.3-7
These are not all the possible side effects of NRTIs. Talk to your doctor about what to expect when taking an NRTI. You also should call your doctor if you have any changes that concern you when taking an NRTI drug.
Other things to know
Before starting treatment with an NRTI drug, tell your doctor if you:3-7
- Have liver problems, including hepatitis
- Have kidney problems or are receiving dialysis
Have bone problems - Have an infection
- Are pregnant, planning to become pregnant, or breastfeeding
NRTIs may interact with other medicines and cause serious side effects. Before beginning treatment for HIV, tell your doctor about all your health conditions and any other drugs, vitamins, or supplements you take. This includes over-the-counter drugs.