Protease Inhibitors
Reviewed by: HU Medical Review Board | Last reviewed: May 2025 | Last updated: May 2025
Protease inhibitors (PIs) are a class of drugs used to suppress HIV in the body. When PIs are used along with other HIV-fighting drugs, the treatment regimen is referred to as antiretroviral therapy (ART).1,2
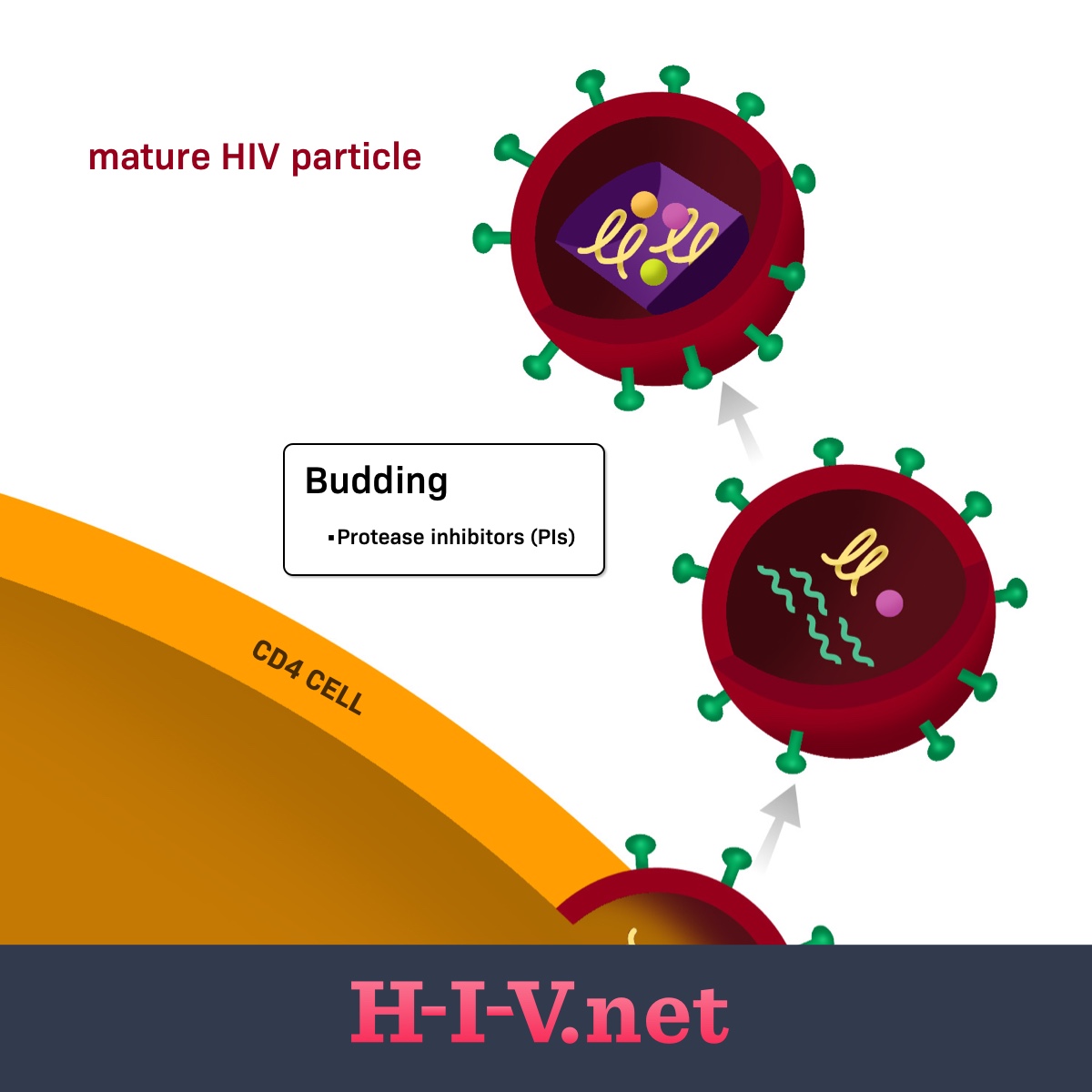
PIs act on the assembly portion of the HIV life cycle, where the virus reassembles itself into new, mature HIV particles after replication. PIs prevent these new, replicated, mature viruses from fully assembling and infecting new cells to start the process over again.1,2
HIV life cycle
Viruses, like HIV, need human host cells to replicate. They cannot multiply on their own without human cells. When HIV particles, called virions, enter the body after a transmission event, the next main steps of the HIV lifecycle are as follows:3
- Binding
- Fusion
- Reverse transcription
- Integration
- Replication
- Assembly
- Budding
Figure 1. Protease inhibitors target budding in the HIV life cycle
The first two steps of the life cycle focus on HIV getting into a human CD4 cell, or T cell, which is its target cell. Once inside, it needs to disassemble itself and begin the replication process.3
The next three steps represent the replication process of the virus’s genetic material. The last two steps of the process involve HIV re-assembling itself into new, mature virions that can be released from the CD4 cell and enter the bloodstream where they can go on to infect new cells.3
Fully formed HIV carries around with it its viral RNA (its genetic material) and several enzymes, including the reverse transcriptase enzyme, the integrase enzyme (both involved in replication), and the protease enzyme. An HIV virion is not considered complete and able to infect more CD4 cells without all of these components.3
How do protease inhibitors work?
PIs prevent newly replicated, mature viruses from being formed and released into the blood to further infect other CD4 cells. HIV’s genetic information comes in 2 forms. The first is its actual genetic information, the virus’s RNA, that it reverse transcribes into DNA through the reverse transcription process. This new DNA gets incorporated into host cell DNA and allows the virus to continue to replicate itself. This incorporated DNA can also get turned into viral mRNA, the second important form of HIV genetic information.1,2,4-7
Messenger RNA (mRNA) is used to create proteins, including the enzymes HIV virions carry around with them to live. During the process of converting mRNA into these proteins, the virus uses its protease enzyme. The protease enzyme cuts the polyprotein made from mRNA at certain sites to create the proteins needed for new virion particles. PIs block the protease enzyme and prevent it from cutting the polyprotein into the proteins needed. Without this cutting or cleavage of the polyprotein, the final protein products aren't able to be made.1,2,4-7
Examples of protease inhibitors
Protease inhibitors approved to treat HIV include:1,2,4-7
- Aptivus® (tipranavir)
- Lexiva® (fosamprenavir)
- Prezista® (darunavir)
- Reyataz® (atazanavir)
What are the possible side effects?
Side effects can vary depending on the specific drug you are taking. The most common side effects of PIs include:4-7
- Abdominal pain
- Nausea, vomiting
- Diarrhea
- Rash
- Cough
- Tiredness
- Kidney stones
- Redistribution of fat on your body (lipodystrophy)
- Changes in how food tastes
These are not all the possible side effects of PIs. Talk to your doctor about what to expect when taking a PI. You also should call your doctor if you have any changes that concern you when taking a PI drug.
Other things to know
Before starting treatment with a PI, tell your doctor if you:4-7
- Have heart problems
- Have liver problems, including hepatitis B or C
- Have phenylketonuria (PKU)Have kidney problems
- Are receiving dialysis
- Have diabetes
- Have hemophilia
- Are pregnant, planning to become pregnant, or breastfeeding
PI medicines can interact with other drugs and cause serious side effects. Before beginning treatment for HIV, tell your doctor about all your health conditions and any other drugs, vitamins, or supplements you take. This includes over-the-counter drugs.